
The \(4f\) sublevel belongs to the sixth period. Typology edit In 1934, George Quam, a chemistry professor at Long Island University, New York, and Mary Quam, a librarian at the New York Public Library complied and published a bibliography of 133 periodic tables using a five-fold typology: I. The \(f\) sublevel is always two levels behind. B Thus beryllium has an He s2 electron configuration. Beginning with beryllium, we see that its nearest preceding noble gas is helium and that the principal quantum number of its valence shell is n 2. In other words, the \(3d\) sublevel fills during the fourth period. Solution: A The group 2 elements are in the s block of the periodic table, and as group 2 elements, they all have two valence electrons. The figure also illustrates how the \(d\) sublevel is always one principal level behind the period in which that sublevel occurs. The periodic table can be divided into different sections. periodic-table-broken-rows-blocks-s-p-d-f-blocks-block-different-. The sublevels are not readily identified on the periodic table, but they are relatively easy to learn. For example, substances like water and copper sulfate are not in the periodic table because they are compounds.\) (Credit: Christopher Auyeung and Joy Sheng Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window)) Blocks in periodic table Study Guide Inspirit Web20 de sept. Only elements are found in the periodic table, never compounds. The periodic table is a tabular arrangement of the chemical elements, ordered by their atomic number, electron configurations, and recurring chemical properties.This ordering shows periodic trends, such as elements with similar behaviour in the same column.It also shows four rectangular blocks with some approximately similar chemical properties. 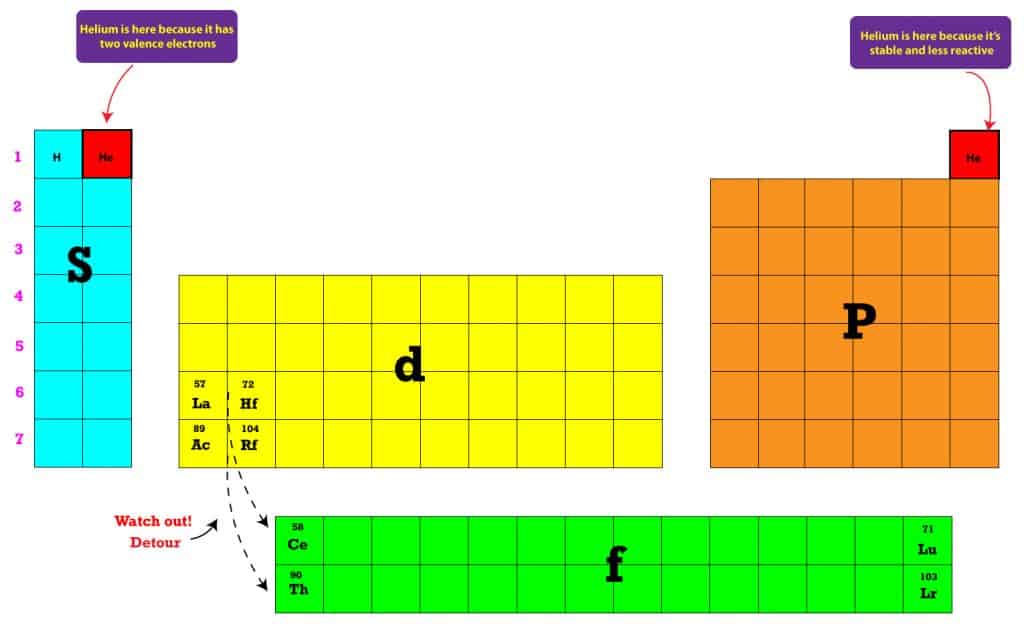
Most elements are metals, rather than non-metals.Įach element has its own chemical symbol, made from letters. Hydrogen is a non-metal but it is often put in the middle. The zig-zag line in this diagram separates the metals, on the left, from the non-metals, on the right. there are only two elements in Period 1 (hydrogen and helium) Periodic Properties of Class 11 s - Block elements: Elements in which the last electron enters the s- orbital or their respective outermost shells are called s.the block in between Group 2 and Group 3 is where the transition metals are placed.the main groups are numbered from 1 to 7 going from left to right, and the last group on the right is Group 0 An alternate numbering system numbers all of the s, p, and d block elements from 1-18.the metals are on the left and the non-metals are on the right.elements in the same group are similar to each other.Each element is placed in a specific location because of its atomic structure. A cut between the halogens and the alkali-metals, where the largest change of chemical behavior occurs, became the most favored one, at least among practicing chemists. Elements as Building Blocks The periodic table is organized like a big grid. All the different elements are arranged in the periodic table. Later inventors of Periodic Tables cut, so to speak, this cylindrical bent surface at different points, in order to obtain flat printable tables. The blocks in the periodic table are majorly divided into four different blocks: the s-block, p-block, d-block, and f-block.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
